
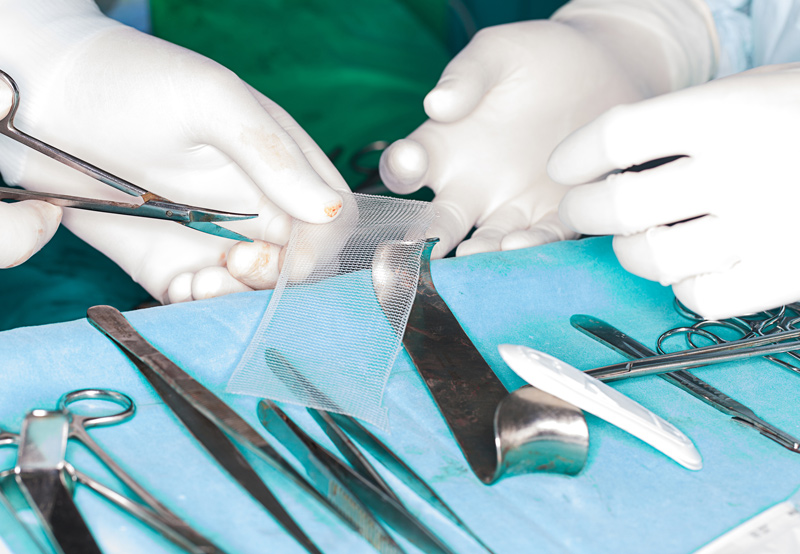
Mesh used to be a minimally invasive, gold standard treatment for stress urinary incontinence (SUI). However, mesh complications have been catastrophic, leading to a range of symptoms and conditions like erosion, pain, voiding dysfunction, infection recurrent UTIs, organ perforation, bleeding, vaginal scarring, neuromuscular alterations, fistulae, lower urinary tract symptoms, bowel complications, and immune disorders.
Research has shown that at least 42% of patients who received mesh for stress incontinence experienced at least one adverse event, and 12% had at least one serious adverse event.
The most common complications of treatment with tapes or slings are pain and mesh exposure (extrusion/erosion), followed by voiding dysfunction, urgency urinary incontinence, recurrent UTI, and SUI.
Between 2.3% and 6.1% of women require intervention for mesh complications. Removal was necessary after one and nine years of mid-urethral sling placement for an estimated 1.4% and 3.3% of women, respectively.
What is Mesh?
Mesh is a synthetic, net-like material made from various types of polymers. Female synthetic slings are typically made from polypropylene, monofilament with macropores mesh since it is less likely to cause a foreign-body response and improves tissue incorporation.

Mesh can be inserted for the following reasons:
- As a synthetic sling to treat women with SUI; includes retropubic lings, transobturator and single-incision mini-slings
- To treat anterior, vault, and posterior compartment prolapse; inserted vaginally. (This mesh is currently banned to be used per FDA regulation)
- To treat uterine/cervical or vault prolapse through sacrocolpopexy and sacrohysteropexy; inserted abdominally
- To treat rectal prolapse through rectopexy
Risk Factors for Mesh Complications
The risk of developing complications from mesh treatment include:
- Older age
- Diabetes mellitus
- Smoking
- Prior pelvic radiation
- Immunosuppression
- Previous vaginal surgery
To reduce the risk of mesh complications, patients need to keep their glucose under control, quit smoking, and maintain a healthy weight.
Diagnosing Mesh Complications
To determine the cause of mesh complications, physicians will consider mesh material factors, patient factors, and surgeon factors.
The physician will conduct a multidisciplinary evaluation, looking at hormonal status, comorbidities such as autoimmune disorders and fibromyalgia, the type of mesh that was used, and surgical details of previous attempts to remove the mesh, such as pictures of the removed mesh and outcomes.
The physician will also ask the patient questions about their symptoms, vaginal discharge or bleeding, mobility issues, urinary symptoms, storage and voiding symptoms, and sexual dysfunction.
A physical, vaginal examination will be done to identify:
- Mesh exposure sites
- Pain trigger points
- Vaginal scarring
- New or recurrent prolapse
- Signs of infection
- Vaginal discharge
- Tender areas
- Previous surgical scars
- Urine retention
Patients may be asked to complete a quality-of-life questionnaire to assess pelvic floor symptoms, sexual function, pain, and quality of life.
In addition, patients experiencing lower urinary tract symptoms will complete a urine test to identify the presence of blood and leukocytes.
Urethrocystoscopy is the preferred tool for diagnosing mesh complications in patients experiencing recurrent UTIs, stone formation, voiding dysfunction, and hematuria.
Other tests that may be done include cystography, CT scan of the pelvis and abdomen, video-urodynamics, translabial/transvaginal ultrasound, and magnetic resonance imaging (MRI).

